 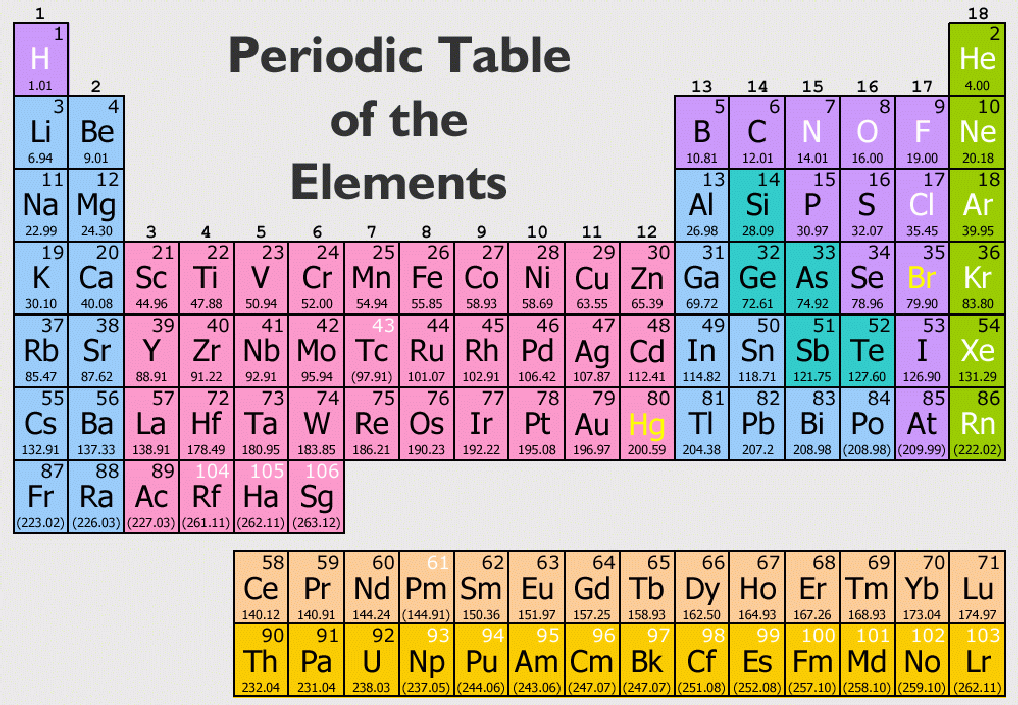 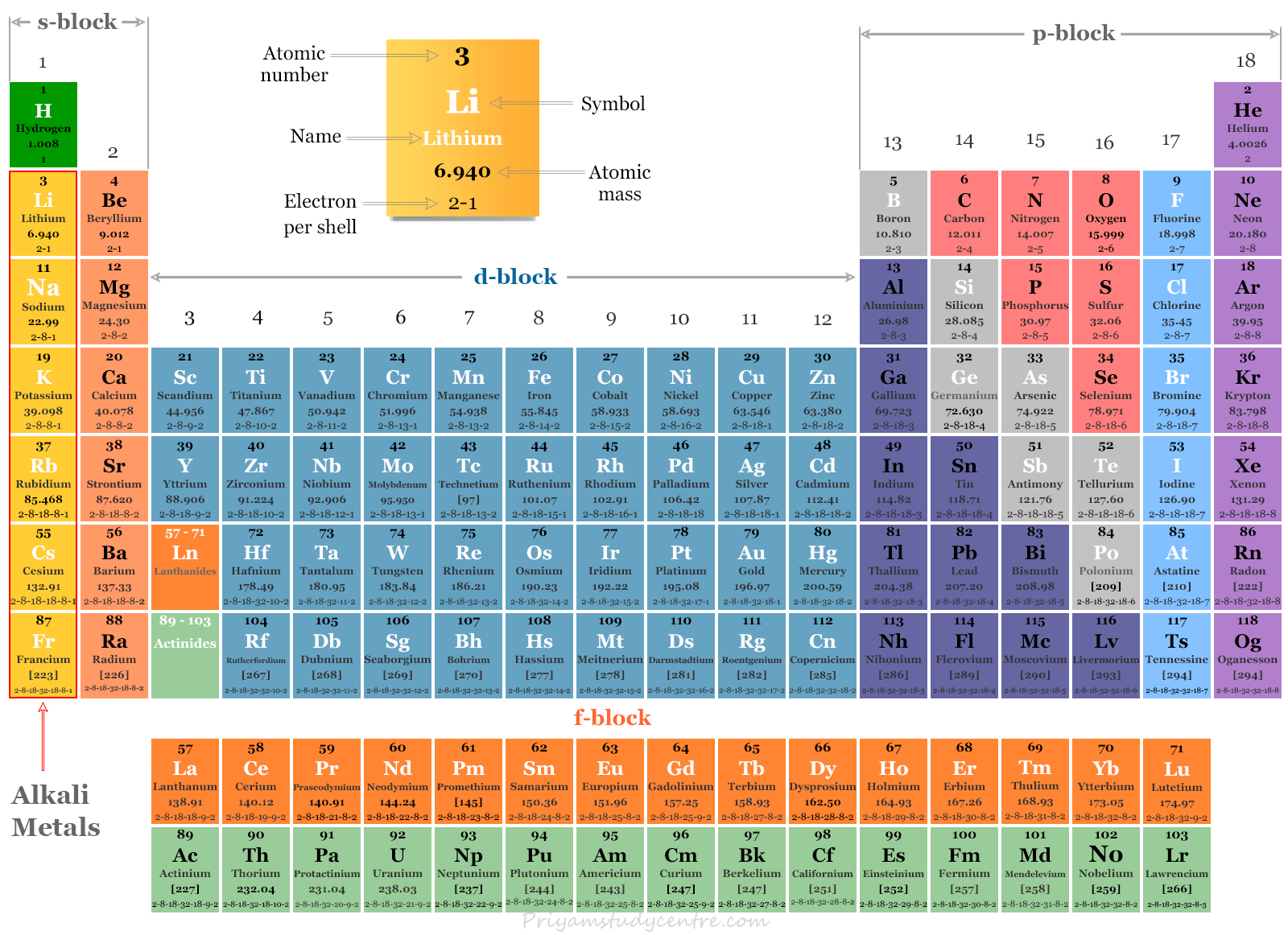
"All of these elements were first discovered in compounds some of the discoveries are hard to attribute due to the abundance and usage of the compounds," says Nataro. If an element is highly reactive, it's harder to find its pure form in nature. For example, sodium chloride (table salt) and sodium carbonate (soda ash) are widely available sodium compounds. Since alkali metals react readily, they are usually found in conjunction with other metals in nature. So, all of the alkali metals like to make cations that have a charge of +1." Alkali Metals in Nature When this happens, the atom is referred to as an ion and since it would have a positive charge, it is called a cation. "As electrons have a charge of -1, losing an electron causes the atom to have a charge of +1. Chip Nataro, chemistry professor at Lafayette College in Easton, Pennsylvania. All of the alkali metals like to give up their single valence electron," says Dr. In this process, the alkali metal is said to be oxidized, and whatever takes the electron from the alkali metal is reduced. "Since the alkali metals only have one valence electron, they typically achieve this state by giving up that electron. That's why chemists talk about atoms wanting to achieve a "noble gas configuration." Noble gases (elements like neon and helium) are not very reactive because their outermost electron shells are full. In fact, reactivity in chemistry is defined by the number of electrons in the outermost shell. Having only one valence electron in the outermost shell makes it very easy for the alkali metal atoms to reach points of stability - they just need to lose one electron! This willingness and ease of losing an electron to reach a state of equilibrium is known as high reactivity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
